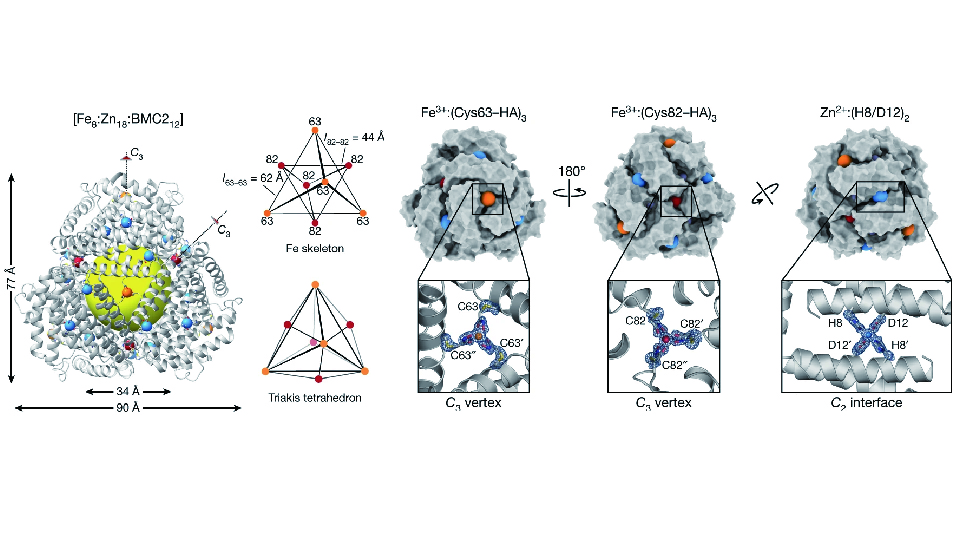
Proteins represent the most versatile building blocks available to living organisms or the laboratory scientist for constructing functional materials and molecular devices.1 The error-free self-assembly of proteins into ordered yet dynamic nano- and microscale architectures, such as 0D virus capsids, 1D cytoskeletal filaments, 2D bacterial surface layers, and 3D cellular crystals is essential for providing the active materials that underlie the responsiveness of the living systems and their ability to adapt to changes in the environment. Underlying the versatility of proteins as building blocks is an immense structural and chemical heterogeneity that renders the programmable self-assembly of proteins an extremely challenging design task. While advances in computational protein design over the last decade have enabled the construction of increasingly sophisticated protein assemblies, such structures are largely static and devoid of the responsiveness/adaptiveness of natural systems. To circumvent the challenge of designing extensive non-covalent interfaces for controlling protein self-assembly, our group has endeavored to use chemical bonding strategies based on fundamental principles of inorganic chemistry and supramolecular chemistry.2 We have shown that appropriately placed metal coordination or reversible covalent bonds can direct the self-assembly of proteins with minimal design, resulting in discrete or infinite, 0-, 1-, 2- and 3D protein architectures that display high structural order over large length scales yet are dynamic, adaptive and possess tunable/predictable physicochemical properties.3-9 When integrated with synthetic polymers, such designed protein assemblies can display high levels of structural adaptiveness and response to external stimuli, while also capable of self-healing and reversible uptake/release of molecular cargo.10,11 Excitingly, self-assembly into extended architectures also renders proteins highly stable,12 such that they can be incorporated into solid-state devices and can perform mechanosensitive functions under harsh, non-aqueous conditions.13
The current goals of the protein materials sub-group are to further expand the chemical design toolkit for protein self-assembly (e.g., through the use host-guest chemistry or surface interactions), to design non-equilibrium protein assemblies powered by light or chemical fuels, and to integrate designed protein materials into natural or artificial cells for control or development of cellular functions (e.g., new sensing modalities, catalytic networks).
——————————————————————————– Selected Publications ——————————————————————————–
1. Zhu, J. et al. Protein Assembly by Design. Chem. Rev. 121, 13701-13796 (2021).
2. Salgado, E. N., Radford, R. J. & Tezcan, F. A. Metal-Directed Protein Self-Assembly. Acc. Chem. Res. (2010).
3. Brodin, J. D. et al. Metal-directed, chemically tunable assembly of one-, two- and three-dimensional crystalline protein arrays. Nat. Chem. 4, 375-382 (2012).
4. Sontz, P. A., Bailey, J. B., Ahn, S. & Tezcan, F. A. A Metal Organic Framework with Spherical Protein Nodes: Rational Chemical Design of 3D Protein Crystals. J. Am. Chem. Soc. 137, 11598-11601 (2015).
5. Bailey, J. B. & Tezcan, F. A. Tunable and Cooperative Thermomechanical Properties of Protein–Metal–Organic Frameworks. J. Am. Chem. Soc. 142, 17265-17270 (2020).
6. Bailey, J. B., Zhang, L., Chiong, J. A., Ahn, S. & Tezcan, F. A. Synthetic Modularity of Protein–Metal–Organic Frameworks. J. Am. Chem. Soc. 139, 8160-8166 (2017).
7. Alberstein, R., Suzuki, Y., Paesani, F. & Tezcan, F. A. Engineering the entropy-driven free-energy landscape of a dynamic nanoporous protein assembly. Nat. Chem. 10, 732-739 (2018).
8. Suzuki, Y. et al. Self-assembly of coherently dynamic, auxetic, two-dimensional protein crystals. Nature 533, 369 (2016).
9. Golub, E. et al. Constructing protein polyhedra via orthogonal chemical interactions. Nature 578, 172-176 (2020).
10. Zhang, L., Bailey, J. B., Subramanian, R. H. & Tezcan, F. A. Hyperexpandable, self-healing macromolecular crystals with integrated polymer networks. Nature 557, 86-91 (2018).
11. Han, K., Na, Y., Zhang, L. & Tezcan, F. A. Dynamic, Polymer-Integrated Crystals for Efficient, Reversible Protein Encapsulation. J. Am. Chem. Soc. 144, 10139-10144 (2022).
12. Brodin, J. D., Carr, J. R., Sontz, P. A. & Tezcan, F. A. Exceptionally stable, redox-active supramolecular protein assemblies with emergent properties. Proc. Natl. Acad. Sci. USA 111, 2897-2902 (2014).
13. Vijayakumar, S. et al. Designed 2D protein crystals as dynamic molecular gatekeepers for a solid-state device. Nat. Commun. 15, 6326 (2024).