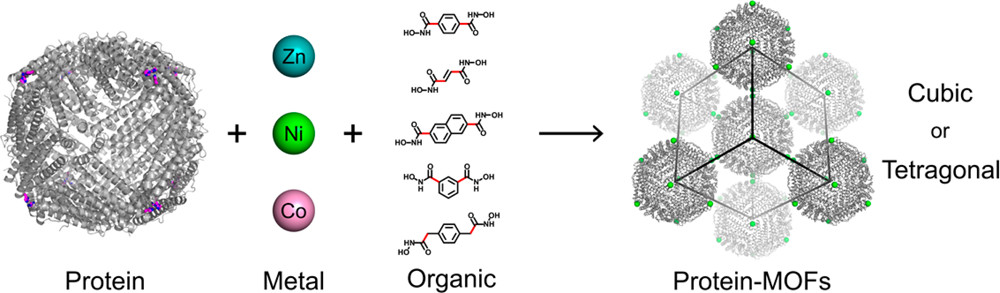
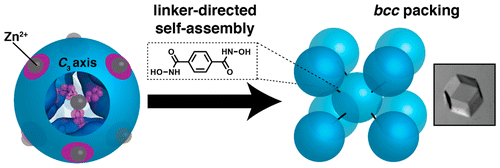
Protein–Metal-Organic-Frameworks
Design of crystalline materials represents a major goal common to numerous scientific disciplines. Proteins are especially attractive building blocks for crystalline materials as they provide high chemical and structural diversity and possess inherent functions such as catalysis, electron transfer, and molecular recognition. Importantly, the formation of ordered, 3D protein crystals is the foundation as well as the rate-limiting step of protein crystallography, providing a strong motivation for their rational design. Although effective methodologies have been developed for facilitating protein crystallization, successes in obtaining 3D protein crystals by design have been rare. In contrast, coordination polymers such as metal-organic frameworks (MOFs) benefit from a high degree of modularity, where component nodes/struts can be interchanged to produce structurally unique architectures with vastly different materials properties. Inspired by MOFs, we developed a method for the formation of crystalline protein arrays based on the construction principles of MOFs, wherein transition-metal loaded spherical protein nodes are interconnected by small ditopic organic linkers possessing metal-binding functional groups. Akin to traditional MOFs, the modularity of these lattices stems from the multiple interchangeable components, which dictate both the crystalline structure and material properties of the protein–MOFs. Research on this new class of materials aims to exploit their highly modular nature, whereby the metal, ligand, and protein scaffold can be systematically varied to assemble unique lattices with different structures and functions.
Principal members: Jake, Ling, Jie
Selected Publications
J.B. Bailey, L. Zhang, J.A. Chiong, S. Ahn, F.A. Tezcan. Synthetic Modularity of Protein–Metal–Organic Frameworks, J. Am. Chem. Soc. (2017).[PDF]
P.A. Sontz*, J.B. Bailey*, S. Ahn, F.A. Tezcan. A Metal Organic Framework with Spherical Protein Nodes: Rational Chemical Design of 3D Protein Crystals, J. Am. Chem. Soc. (2015).[PDF]
Metal-Organic-Frameworks
Metal-organic frameworks (MOFs) are a class of porous material with potential applications in gas storage, separations, drug delivery, catalysis, and chemical sensing. Despite numerous advantages, applications of many MOFs are ultimately limited by their stability under harsh conditions. A common strategy to design stable MOFs is mainly based on Pearson’s hard/soft acid/base (HSAB) theory, where the strong metal-ligand bond facilitates the thermodynamic stability of MOFs. Chelating ligands, such as catecholates and hydroxamates, are well known for their ability to form strong bonds with a wide range of metals. Although living organism such as bacteria uses siderophores (containing hydroxamate or catecholate groups) to effectively assimilate iron in solution, those chelating ligands have been rarely explored in designing stable MOFs. Inspired by nature, we developed a series of hydroxamate-based frameworks, namely HAFs. The designed frameworks demonstrated high chemical stability under harsh conditions, which promote those porous materials great matrix for various applications such as dye uptake, catalysis.
Principal members: Jie
Selected Publications
J.A. Chiong*, J. Zhu*, J.B. Bailey*, M. Kalaj, R. Subramanian, W. Xu, S.M. Cohen, F.A. Tezcan. An Exceptionally Stable Metal-Organic Framework Constructed from Chelate-based Metal-Organic Polyhedra, J. Am. Chem. Soc. (2020).[PDF]